Ferrite materials
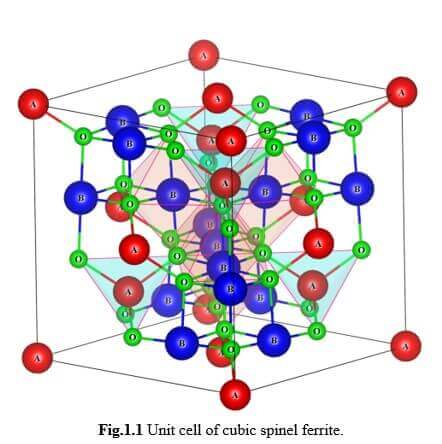
One can find applications of ferrite materials from a simple permanent magnet to most sophisticated equipments in medical fields. Ferrites have an important advantage over other magnetic materials. The oxygen ions in the ferrites insulate the metal ions in the compounds thereby increase the resistivity which leads to decrease in the eddy current losses. High frequency devices, used in several applications, require materials with low eddy current loss and hence, ferrite materials attracted researchers for the last three decades and still continue to do so. Moreover the magnetic property of ferrites can be “tailored” by varying the distribution of cations over the available tetrahedral (A) site and octahedral (B) site in the unit cell. The cations, in micrometer regime have their “own” preferred sites (A or B sites) of occupation in the unit cell, whereas in nanometer regime, this occupation constraint is not strictly adhered and hence, cation distribution can be “tailored”. Methods of preparation and dopants play a major role in bringing down the particle size to nano scale.
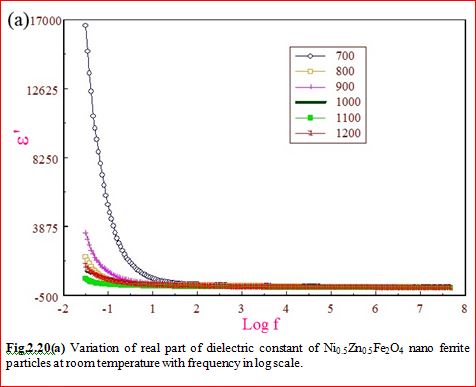
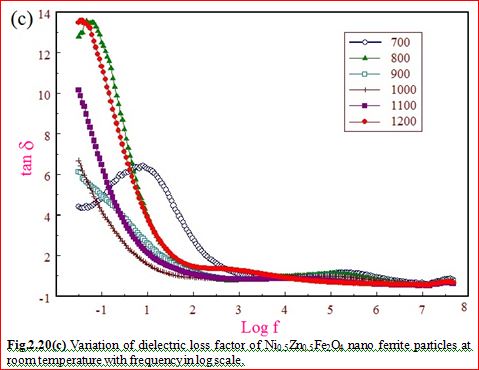
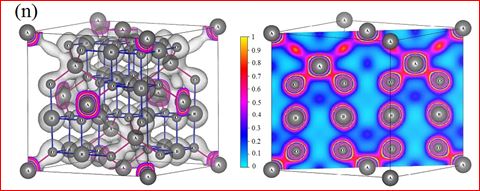
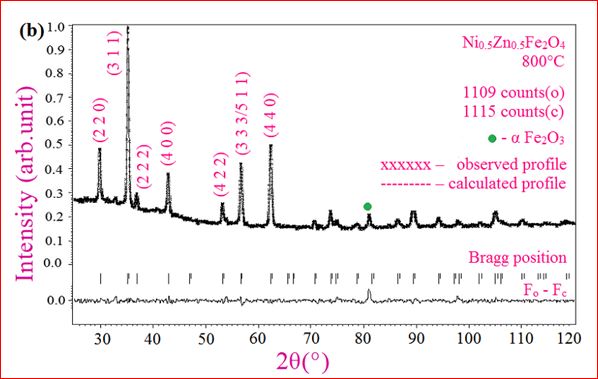
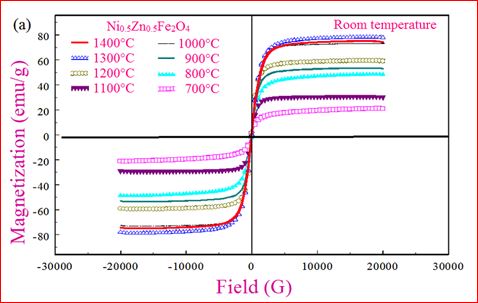
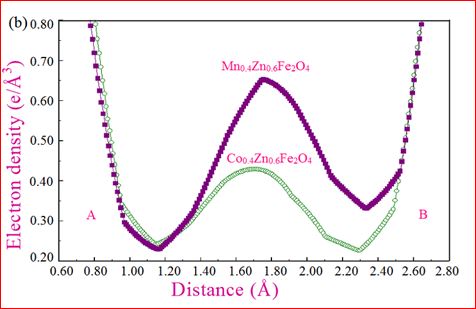
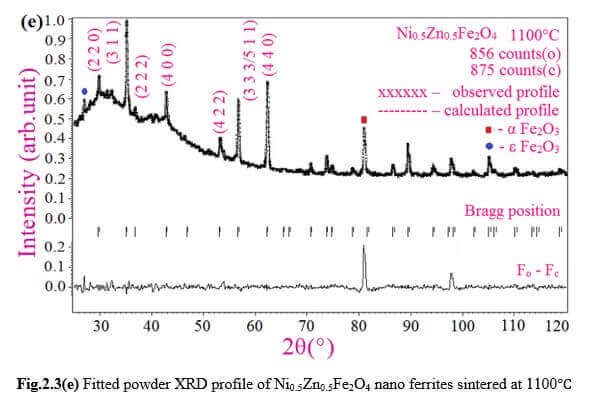
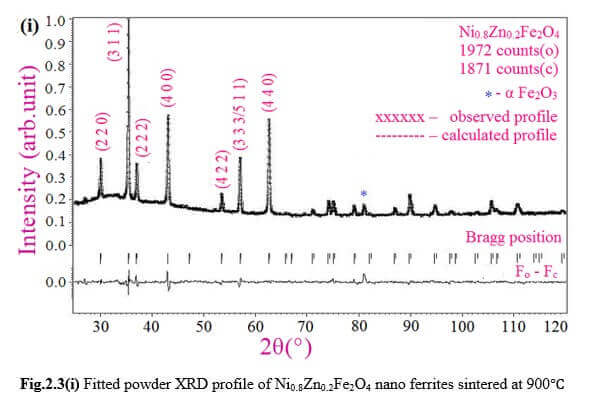
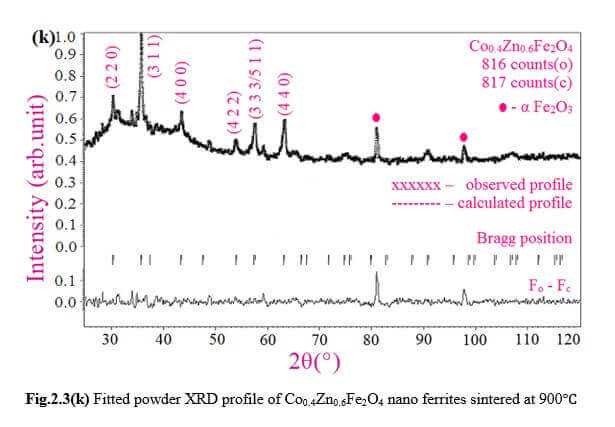
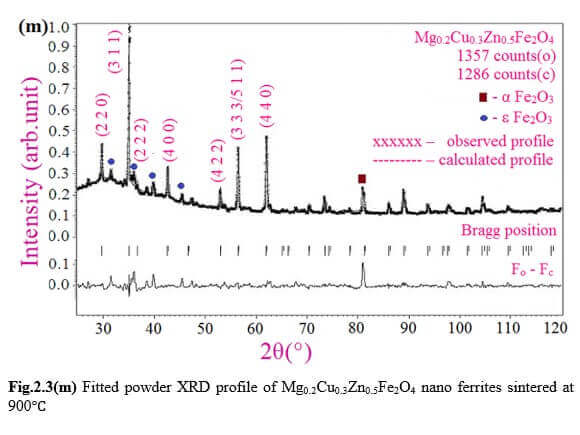
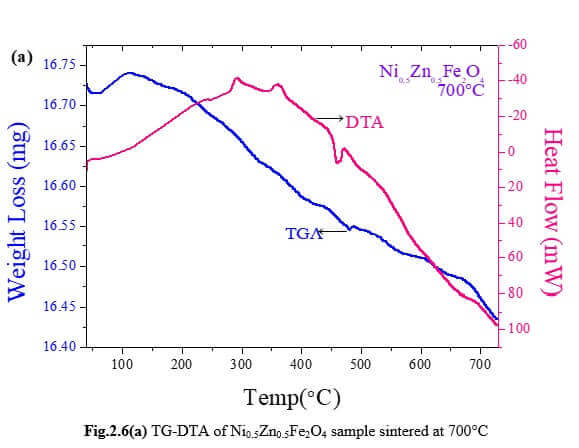
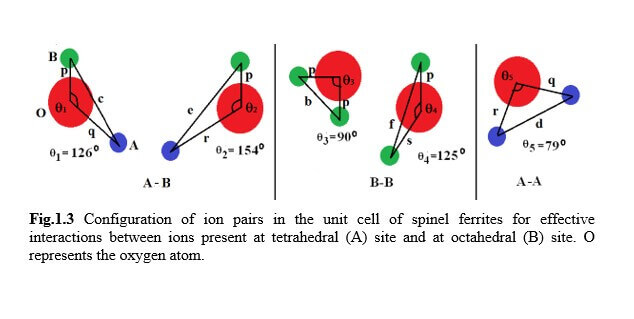
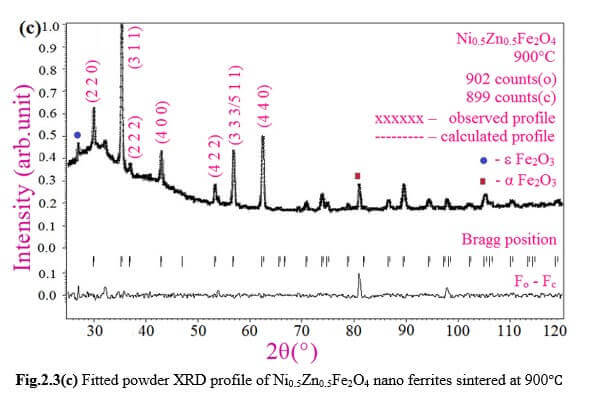
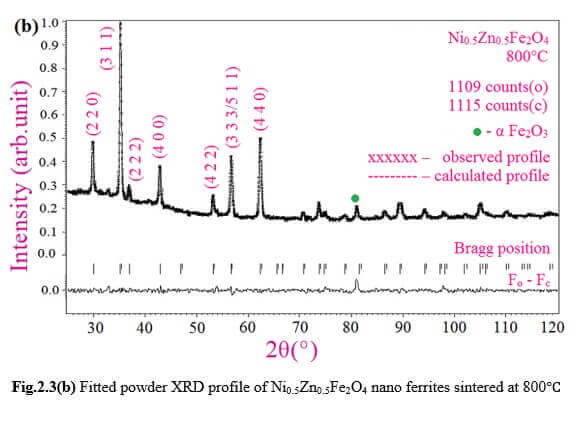
Understanding the bond strengths requires an intense knowledge on charge density distribution. There is not much work has been reported so far on bond strengths between atoms at A-site and B-site of ferrites by deploying maximum entropy method (MEM) techniques in the unit cell of ferrites. This motivated researchers to carry out intense research on the bond strengths and other characterization on ferrites. The physical and chemical properties of atoms or molecules mostly depend on the charge density distribution, bond strength etc. Hence, the following samples were synthesized and studied for their bond strength between various sites (A-A, A-B and B-B sites) using MEM.Ni0.5Zn0.5Fe2O4.
Research Materials
Ni0.5Zn0.5Fe2O4 - sintered at 700°C to 1400°C.
Mn0.4Zn0.6Fe2O4
Co0.4Zn0.6Fe2O4
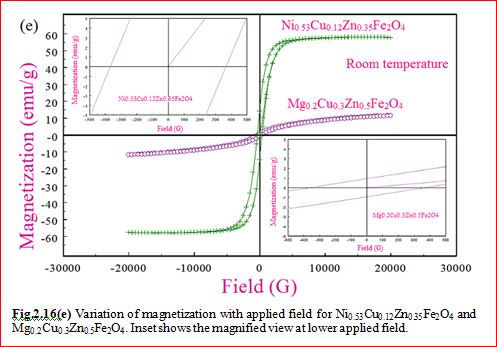
Ni0.53Cu0.12Zn0.35Fe2O4
Mg0.2Cu0.3Zn0.5Fe2O4
Ni0.8Zn0.2Fe2O4 and
NiFe2O4