Multiferroic materials:
The coexisting states of coupled magnetic and ferroelectric ordering in multiferroic materials makes them interesting for the researchers. The material lanthanum orthoferrite (LaFeO3) is one of the most important multiferroic material due to the simultaneous presence of coupled (ferro/antiferro) magnetic and (ferro/antiferro) electric ordering in that system. LaFeO3 is a ABO3 – typed perovskite like structure, which crystallizes in an orthorhombic phase at room temperature. The perovskite oxide LaFeO3 is known to be a canted G-type wide-gap antiferromagnetic insulator at room temperature with high Néel temperature (TN~740°C). In LaFeO3, La3+ ions are at the corners of the unit cell and the Fe3+ ion lies at the centre of octahedron formed by six O2- ions.
The structure, size, physical and chemical properties of LaFeO3 are strongly influenced by the choice of synthesis method along with the substitution of divalent or trivalent or tetravalent ions at the La site of host lattice in LaFeO3. Since LaFeO3 exhibits significant physical and chemical properties, it is of great importance to be used in many branches of modern technologies such as solid oxide fuel cells, photocatalyst, chemical sensors, non-volatile magnetic memory devices, ultrasensitive magnetic read heads of modern hard disk drives, novel spintronic devices, and multistate memories etc.,. To understand the physical and chemical properties of LaFeO3, the electronic distribution and the nature of chemical bonding between the atoms in the unit cell are essential. Therefore, this research work is aimed at synthesis, characterization and charge density distribution analysis of pure and doped LaFeO3 multiferroic materials such as i) La1-xAlxFeO3 ii) La1-xCexFeO3 iii) La1-xSrxFeO3 and iv) La1-xZnxFeO3 . The multiferroics La1-xAlxFeO3, La1-xCexFeO3, La1-xSrxFeO3 have been synthesized through high temperature solid state reaction method and La1-xZnxFeO3 has been synthesized by chemical co-precipitation method.
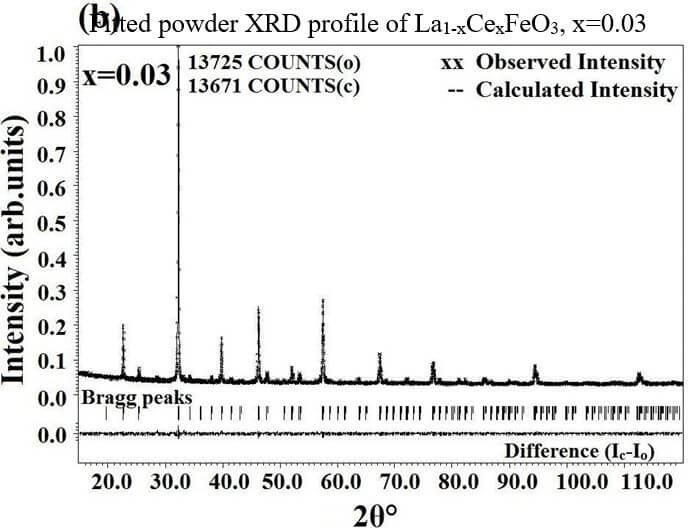
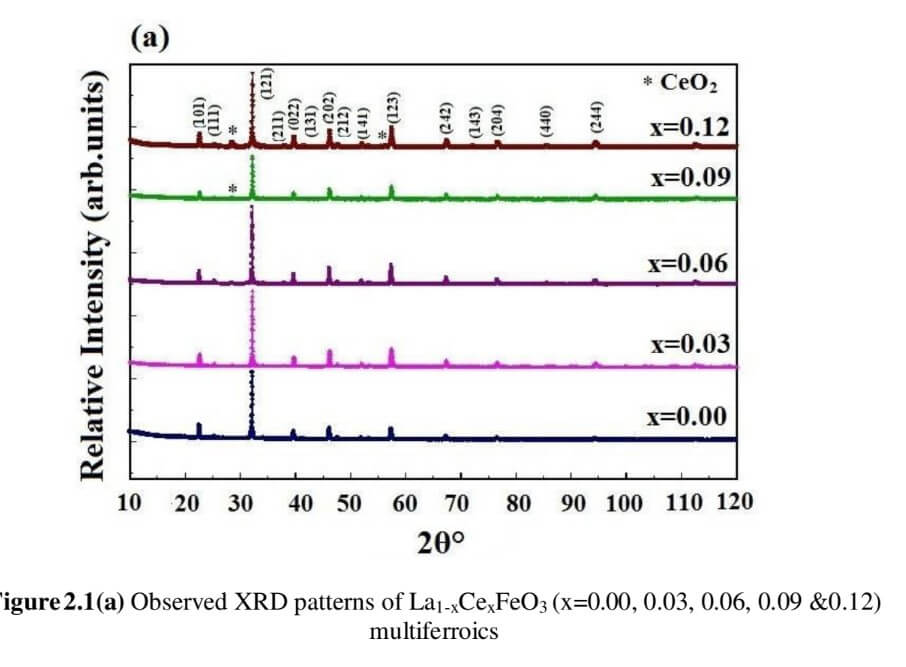
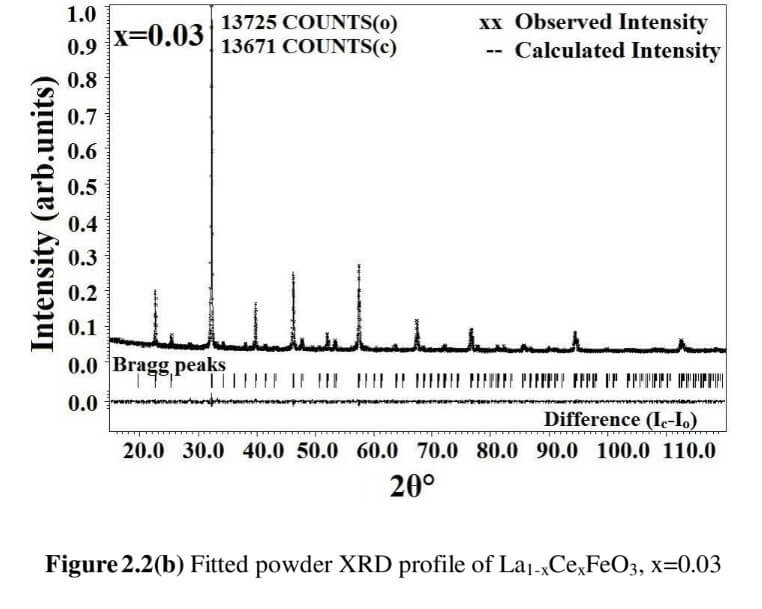
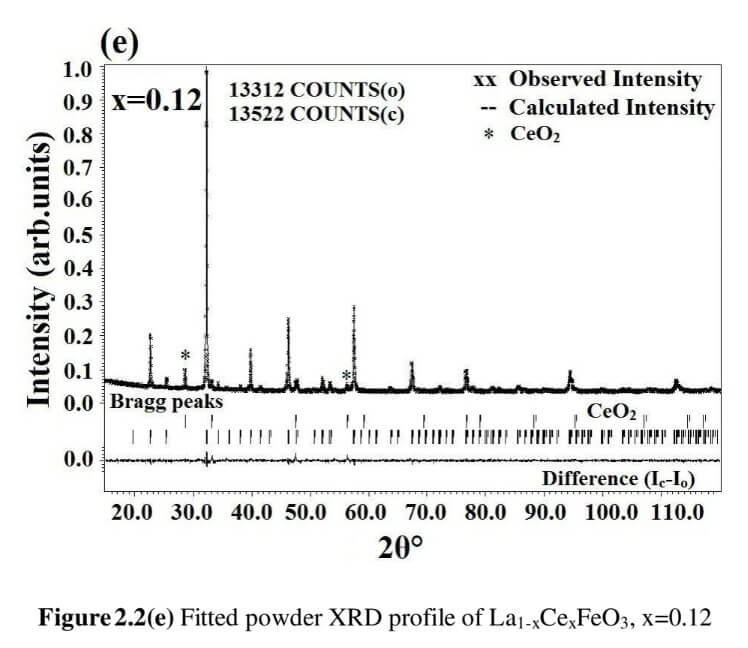
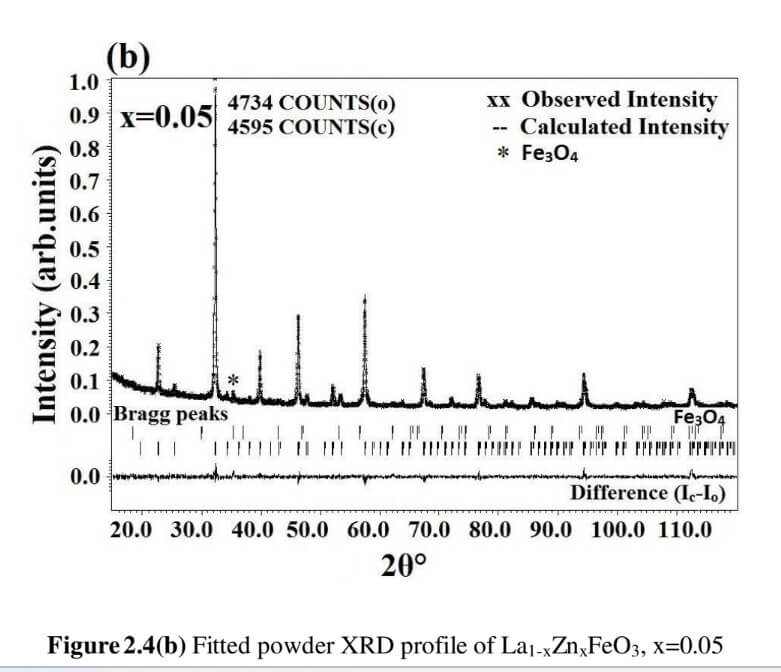
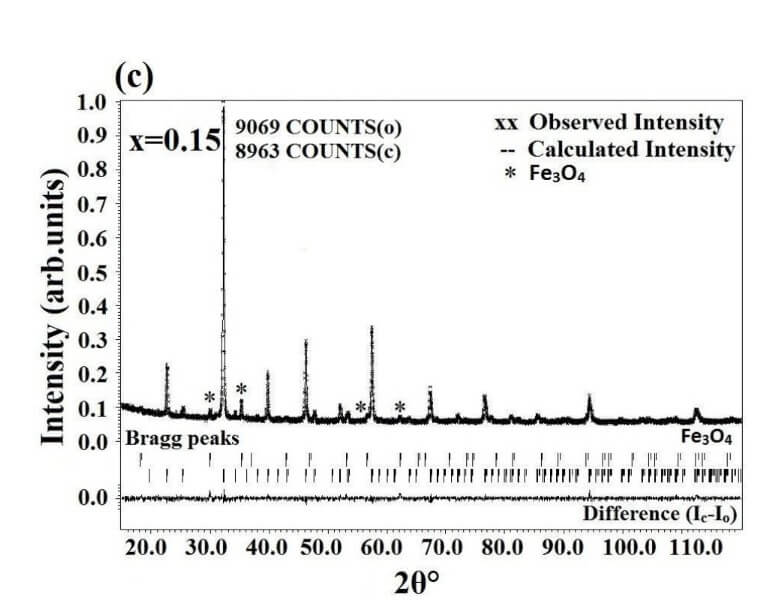
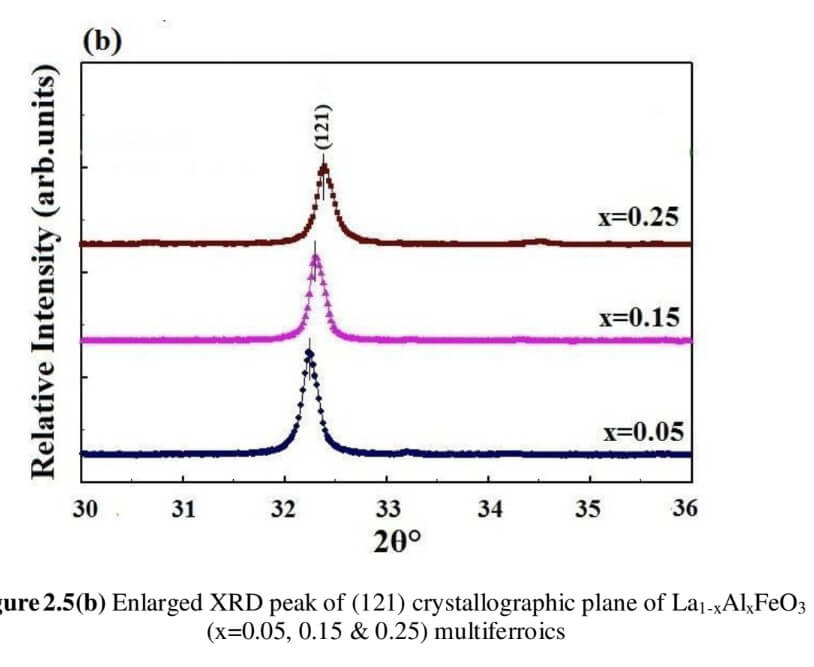
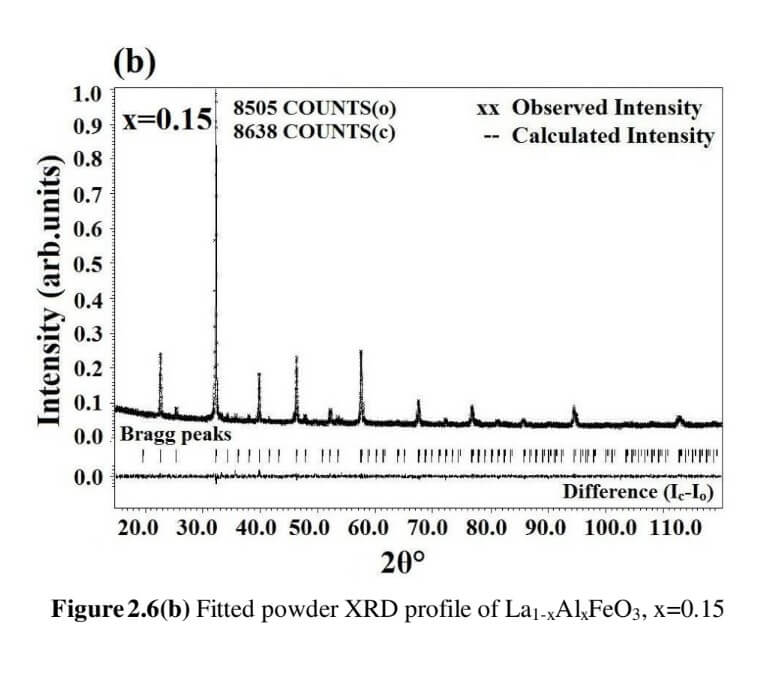
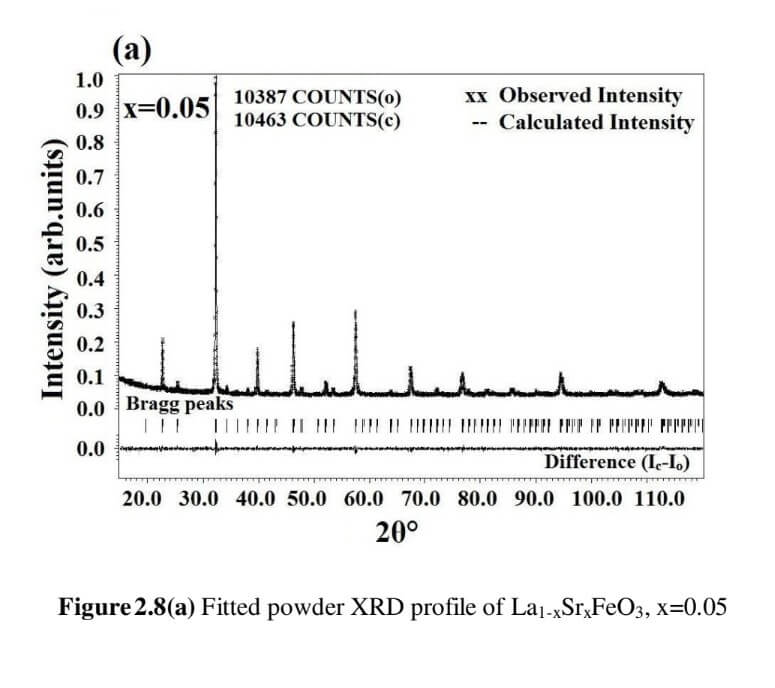
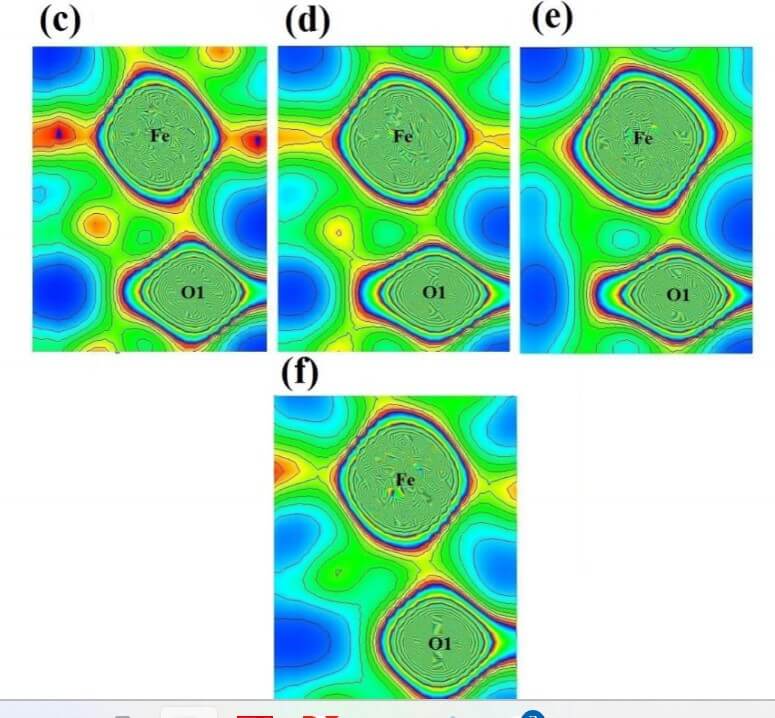
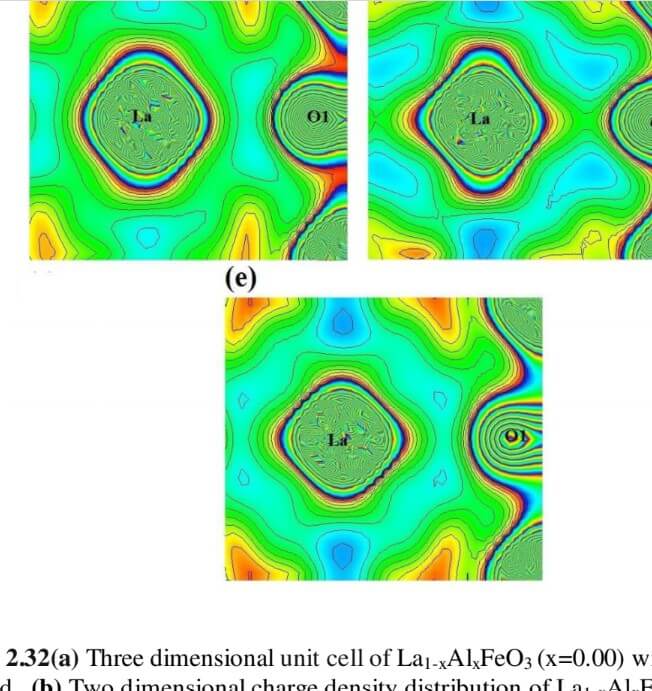
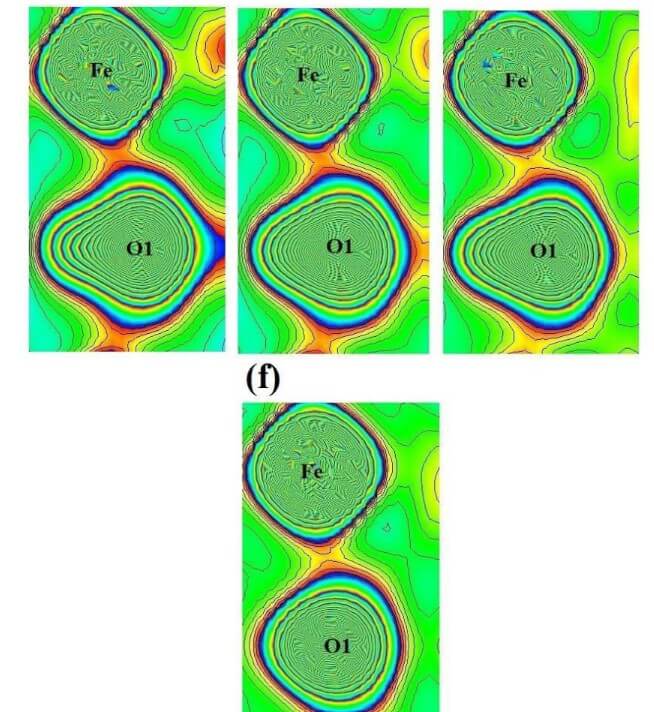
The structural analysis of the synthesized multiferroics has been done through Rietveld refinement process using JANA2006 software. The charge density and interatomic bonding analysis have been done through high resolution maximum entropy method (MEM) by employing the softwares PRIMA and VESTA. The optical behaviour, polarization behaviour, magnetic ordering and electric ordering have been correlated separately with the experimental charge densities which have not been done so far.
Materials of Interest:
Ceramic materials, multiferroic materials, dielectric and magnetic materials
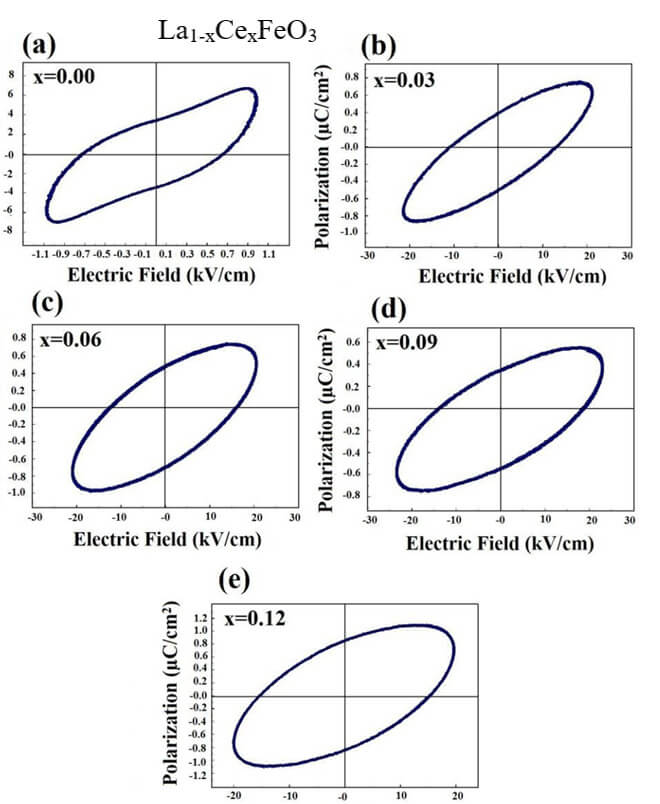
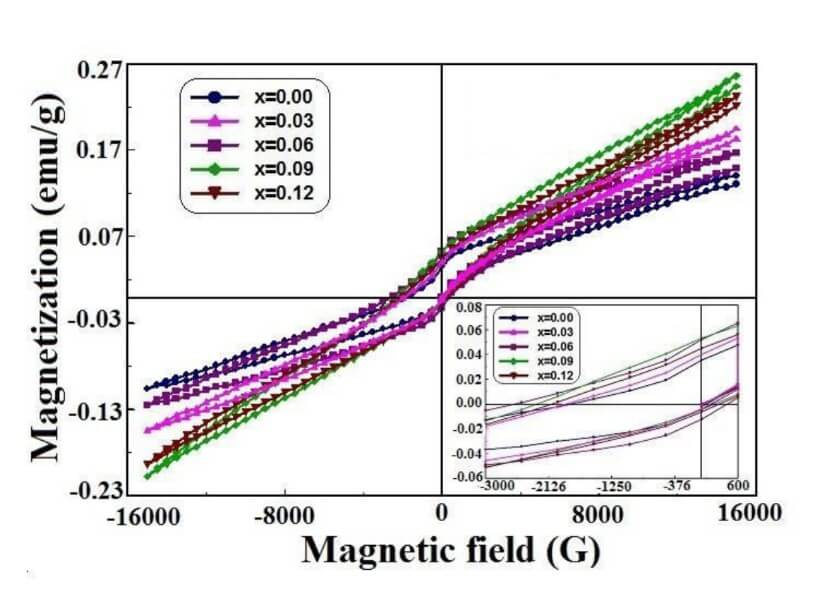
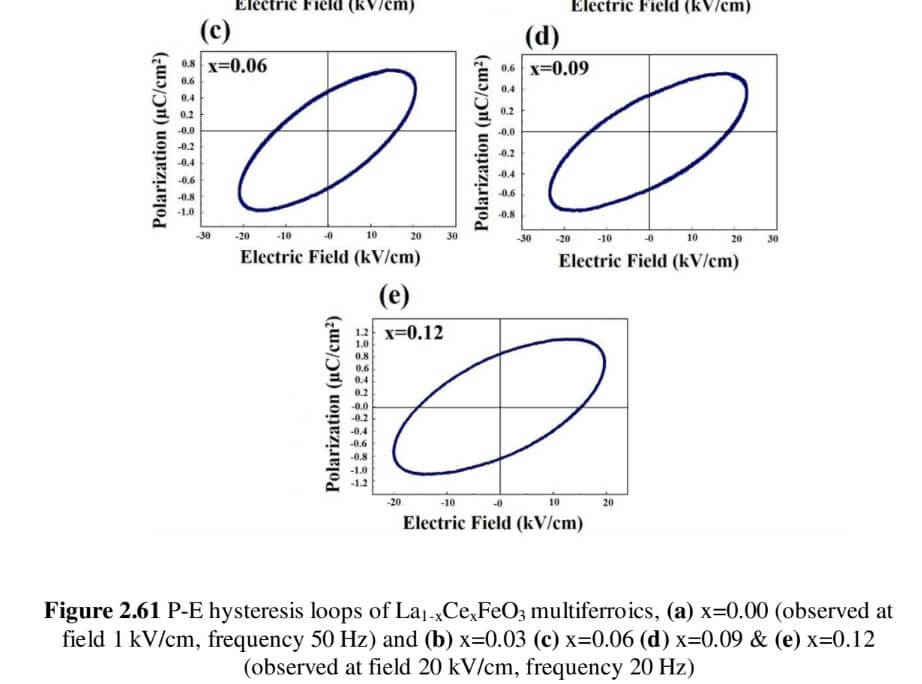
La1-xCexFeO3 (x=0.00, 03, 0.06, 0.09 and 0.12) (LCFO)
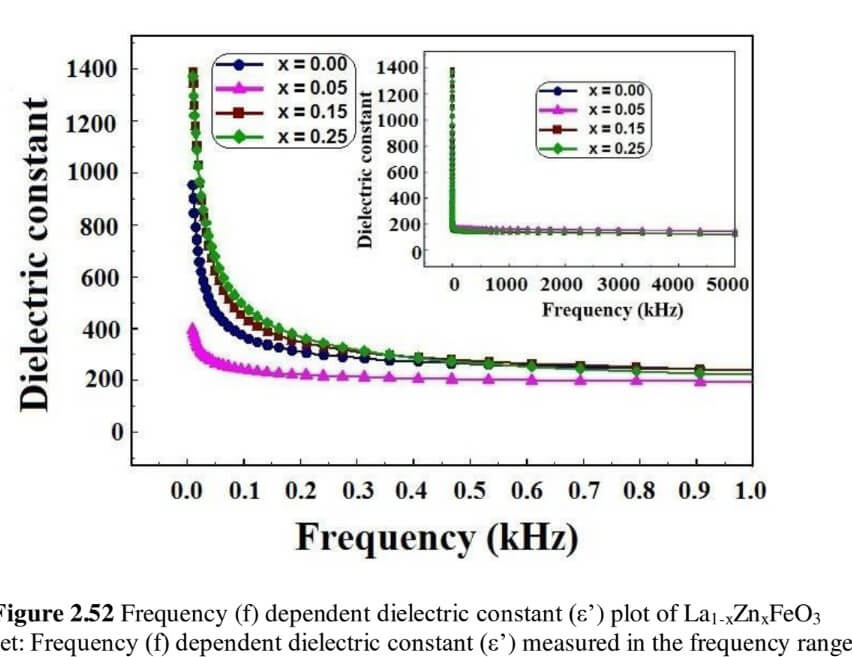
La1-xZnxFeO3 (x=0.00, 0.05, 0.15 and 0.25) (LZFO)
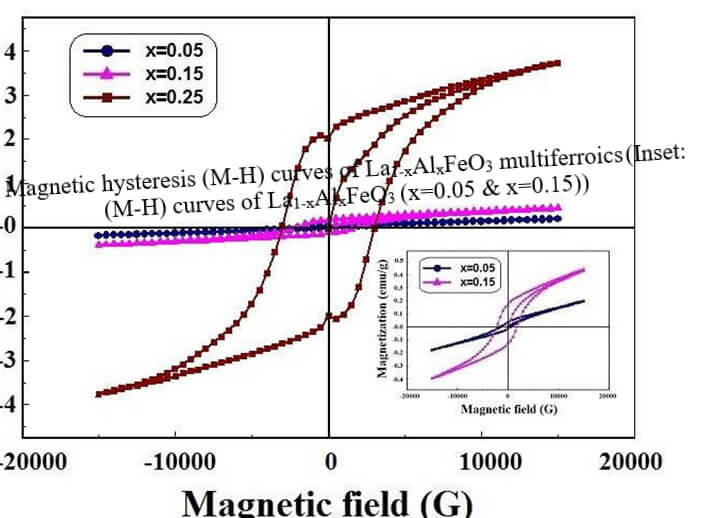
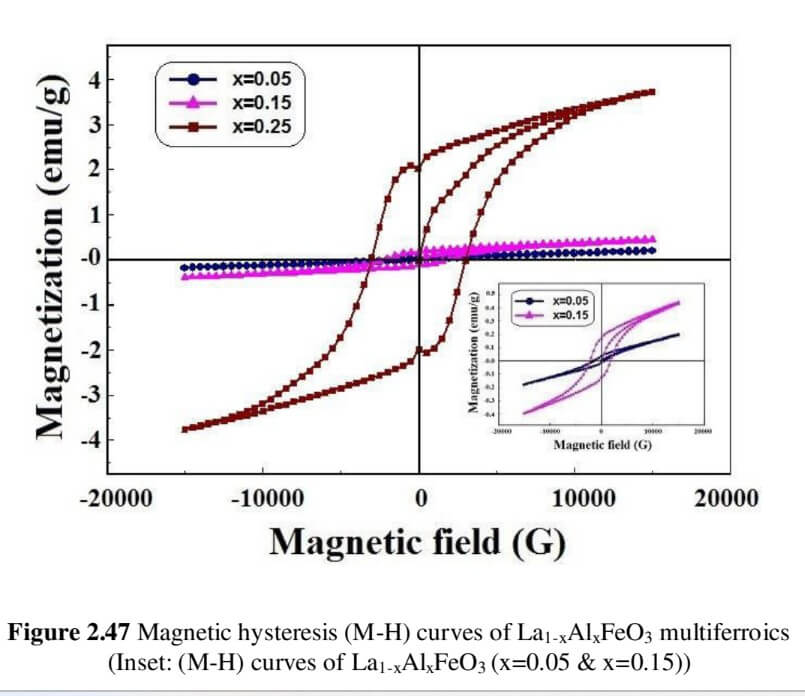
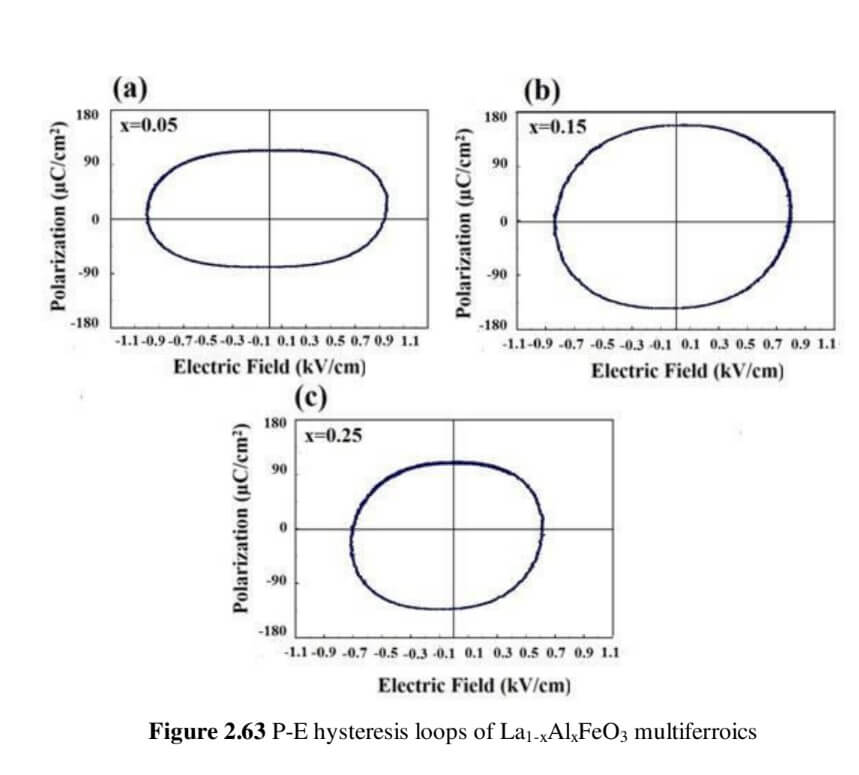
La1-xAlxFeO3 (x=0.05, 0.15 and 0.25) (LAFO)
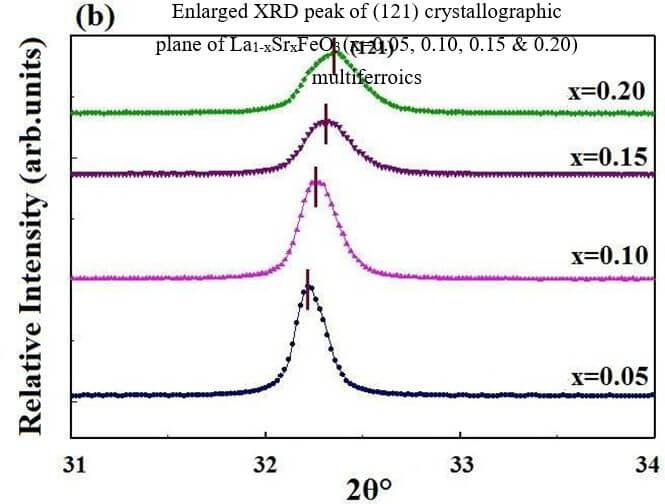
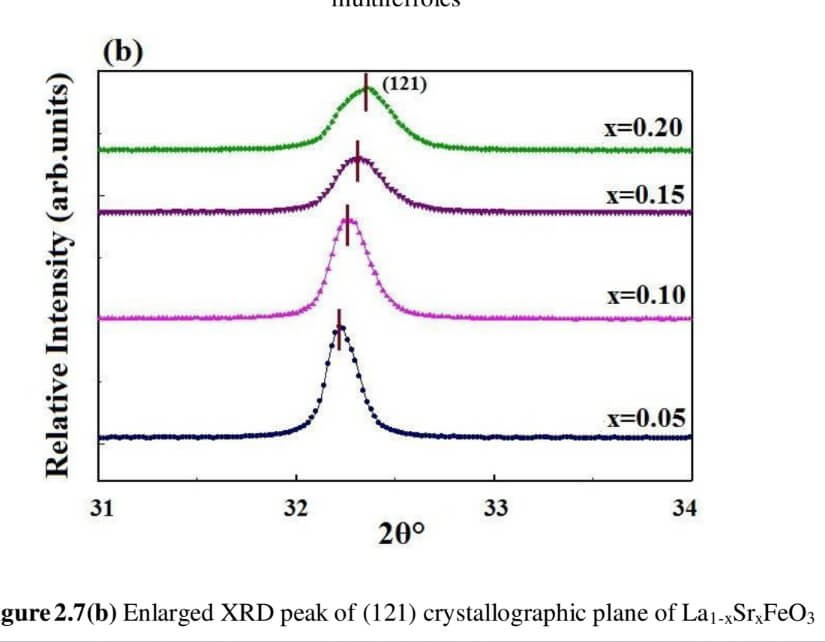
La1-xSrxFeO3 (x= 0.05, 0.10, 0.15 and 0.20) (LSFO)
Viva Photos